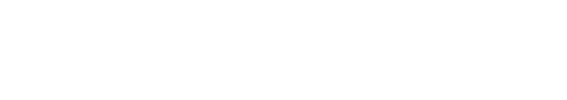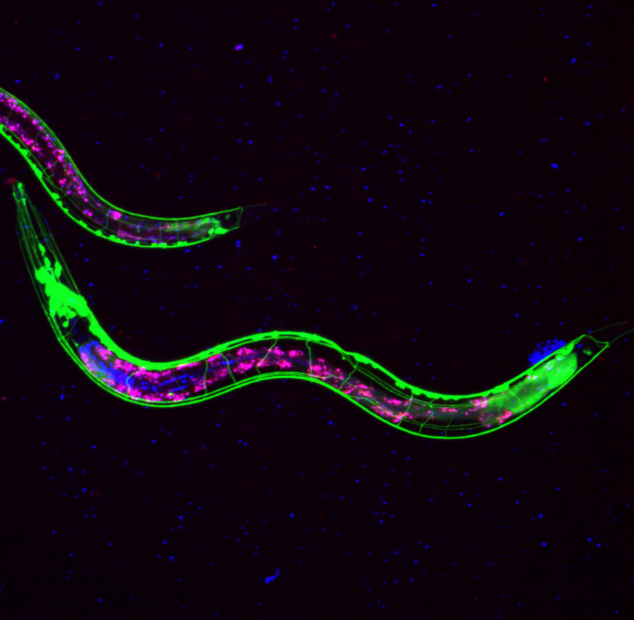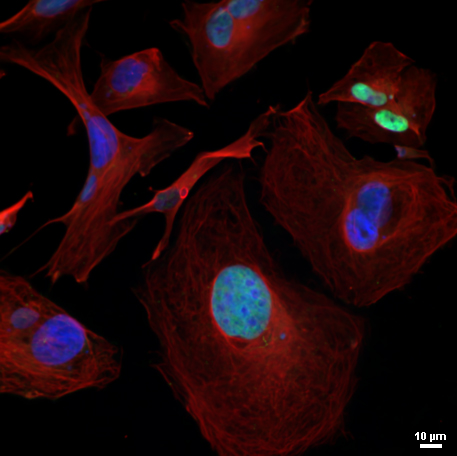
Credit: NeuroWise Laboratory of Environmental Toxicology, University of Toledo
Mote’s Marine Biomedical Research Program uses basic and applied research techniques to study interactions between human and marine health. Research areas include toxicology, pharmacology, gerontology, neuroscience, and immunology. Our primary research efforts use interdisciplinary approaches to 1) investigate how early life exposures to brevetoxins may lead to disease development later in life, 2) explore the potential of marine animal venoms in treating neurodegenerative diseases, and 3) study the remarkable lifespan of marine animals.
Research Description & Primary Goals
- Brevetoxins are produced by red tides (Karenia brevis) and are known to affect the digestive, respiratory, and nervous system in exposed humans. Here, we are interested in studying persistent toxicities and how early-life exposure to brevetoxins may contribute to disease development later in life. In the Marine Biomedical Research Program, our goal is to understand how brevetoxins contribute to chronic diseases and improve the health of humans exposed to brevetoxins.
- Neurodegenerative diseases are among the most debilitating and life-altering chronic diseases. In the United States, estimates suggest that 13.8 million people will be living with Alzheimer’s disease by 2050 and long-term-care for individuals suffering from this disease is expected to cost $1.1 trillion that same year. However, we still lack effective treatment options for neurodegenerative diseases, such as Alzheimer’s disease. Repurposed animal venoms (e.g., snakes, bees) have become a promising avenue for developing new therapies, with evidence suggesting they may improve proteostasis, improve mitochondrial function, and reduce inflammation. In the Marine Biomedical Research Program, we are interested in expanding upon this field by investigating the therapeutic potential of marine animal venoms (e.g., rays, lionfish) in neurodegenerative diseases. This work may help identify new treatment strategies for these destructive diseases and guide future drug development studies.
- We are also interested in the incredible lifespan of marine animals and the ability of these animals to avoid “aging.” One example of a marine animal with an incredible lifespan is the jellyfish species, Turritopsis dohrnii. The maximum lifespan of T. dohrnii (AKA the “immortal jellyfish”) has never been recorded and this species has shown the capability to revert from an adult to juvenile developmental state, making it an interesting species in the world of gerontology – or the study of aging. Whales are another interesting marine animal, as they are capable of evading at least one “Hallmarks of Aging” – or biological changes that naturally accumulate in the body with age. Whales exhibit an incredible resiliency to genomic instability, one of the strongest factors in biological aging, but mechanisms by which whales evade genomic instability are unclear. Understanding how the “immortality” of species such as T. dohrnii or the ability of animals such as whales to evade hallmarks of aging may help us understand how to slow the biological aging process in humans.
Biomedical & Technology Development at Mote

Marine Biomedical Research Program Team
Additional Program Information
Awards & honors to program staff
Dr. Sam Vielee
-
Graduate Intern Fellowship Training Award, Society of Toxicology (2023)
-
NIEHS T32 Predoctoral Fellowship (2023 – 2025)
-
1st Place Graduate Student Research Award, Society of Toxicology Metals Specialty Section (2025)
-
Journal Cover Photo for NeuroToxicology‘s January 2025 Issue (Volume 106) (2025)
-
Dissertation Completion Award, University of Louisville Graduate School (2025)
-
Graduate Dean’s Citation, University of Louisville Graduate School (2025)
-
John Richard Binford Memorial Award, University of Louisville Graduate School
Grants
Project Title: Shark Immune Cell-Derived Peptides as Novel Agents for Cancer Therapy
Granting Organization: Henry L. and Grace Doherty Charitable Foundation, plus matching support from numerous private foundations, charitable trusts, and private individuals
Project PI’s: Carl Luer, Cathy Walsh
Project Title: Tumor Cell Inhibitory Activity of Compounds Isolated from Culture Medium of Shark Immune Cells
Granting Organization: Florida High Tech Corridor Industry Seed Grant Program
Project PI’s: Carl Luer, Cathy Walsh
Project Title: Novel Compounds from Stingray Epidermal Mucus With Antimicrobial Activity Against Wound Infection Pathogens
Granting Organization: U. S. Department of Defense
Project PI’s: Carl Luer, Cathy Walsh, Kimberly Ritchie, AB Bodine, Laura Edsberg, Jennifer Wyffels, Andrew Cannons, Vicki Luna
-
-
Vielee, S. T. and Wise, J. P. (2023). Among Gerontogens, Heavy Metals are a Class of Their Own: A Review of the Evidence for Cellular Senescence. Brain Sci, 13(3), 500. https://doi.org/10.3390/
brainsci13030500 -
Vielee, S. T., Isibor, J., Buchanan, W. J., Roof, S. H., Patel, M., Meaza, I., Williams, A., Toyoda, J. H., Lu, H., Wise, S. S., Kouokam, J. C., Young Wise, J. L., Abouiessa, A., Cai, J., Cai, L., and Wise, J. P. (2024). Employing a Toxic Aging Coin Approach to Assess Hexavalent Chromium (Cr[VI])-Induced Neurotoxic Effects on Behavior: Heads for Age Differences. Toxicol. Appl. Pharmacol., 489, 117007. https://doi.org/10.1016/j.
taap.2024.117007 -
Vielee, S. T., Isibor, J., Buchanan, W. J., Roof, S. H., Patel, M., Meaza, I., Williams, A., Toyoda, J. H., Lu, H., Wise, S. S., Kouokam, J. C., Young Wise, J. L., Abouiessa, A., Cai, J., Cai, L., and Wise, J. P. (2024). Female Rat Behavior Effects from low Levels of Hexavalent Chromium (Cr[VI]) in Drinking Water Evaluated with a Toxic Aging Coin Approach. Appl. Sci., 14(14), 1-26. https://doi.org/10.3390/
app14146206 -
Vielee, S. T., Buchanan, W. J., Roof, S. H., Kahloon, R., Evans, E., Isibor, J., Patel, M., Meaza, I., Lu, H., Williams, A., Kouokam, J. C., Wise, S. S., Guo, L., Wise, R. M., Wise, J. L., Cai, L., Cai, J., and Wise, J. P. (2024). Chromium Selectively Accumulates in the Rat Hippocampus After 90-Days Drinking Water Exposure and Induces Age- and Sex-Dependent Metal Dyshomeostasis. Toxics, 12(10), 722. https://doi.org/10.3390/
toxics12100722 -
Meaza, I., Wise, J. L., Wise, S. S., Lu, H., Williams, A. R., Delnicki, M., Easley, J., Kouokam, J. C., Wise, J. P., Vielee, S. T., Wise, T. F. J., Wise, R. M., and Wise, J. P. (2025). Oropharyngeal Aspiration of Particulate Hexavalent Chromium Increases Chromium Levels in the Lung and Liver, and Induces Essential Metal Dyshomeostasis in the Lung, Liver, and Blood. J. Trace Elem. Med. Bio. 91, 127705. https://doi.org/10.1016/j.
jtemb.2025.127705 -
Griffin, G., Delnicki, M. E., Lu, H., Meaza, I., Brownell, A. R., Vielee, S. T., Bolatimi, O. E., Wise, R. M., Rui, L., Kouokam, J. C., Wise, S. S., Wise, J. P., Cave, M. C., Wise, J. P., and Wise, J. L. (2025). Exposure to Current Regulatory Levels of Hexavalent Chromium in Drinking Water Alters Diet-Induced Steatotic Liver Disease in Male Rats. J. Trace Elem. Med. Bio., 91, 127731. https://doi.org/10.1016/j.
jtemb.2025.127731 -
Vielee, S. T., Chen, P., Deza Ponzio, R., Meaza, I., Aschner, M. A., and Wise, J. P. (2025). Hexavalent Chromium Neurotoxicity in Caenorhabditis elegans Targeted GABAergic, Cholinergic, and Dopaminergic Neurons, and Contributed to an Aged Phenotype. NeuroToxicology, 111, 103325. https://doi.org/10.1016/j.
neuro.2025.103325 - Luer, C. A. and K. P. Wong. 1978. Altered erythrocyte membrane proteins in sickle cell patients associated with the severity of the disease. Biochem. Med. 19:95 107.
- Luer, C. A. and K. P. Wong. 1978. The effects of pH and temperature on the circular dichroism of human erythrocyte membranes. Biophysi¬cal Chem. 9:15 22.
- Luer, C. A. and K. P. Wong. 1979. The conformation of Escherichia coli ribosomal protein L7/Ll2 in solution: hydrodynamic, spectro¬scopic, and conformation prediction studies. Biochemistry 18:2019 2027.
- Luer, C. A. and K. P. Wong. 1980. The conformational stability of ribosomal protein L7/Ll2: The effects of pH, temperature and guani¬dinium chloride. Biochemistry 19:176 183.
- Bodine, A. B., C. A. Luer and S. Gangjee. 1984. Determination of cerulo¬plasmin and other copper transport ligands in the blood sera of the nurse shark and clearnose skate. Comp. Biochem. Physiol. 77B:779 783.
- Luer, C. A. and P. W. Gilbert. 1985. Mating behavior, egg deposition, incubation period, and hatching in the clearnose skate, Raja eglanteria. Env. Biol. Fish. 13:161 171.
- Bodine, A. B., C. A. Luer and S. Gangjee. 1985. A comparative study of monooxygenase activity in elasmobranchs and mammals: activation of the model pro carcinogen aflatoxin B1 by liver preparations of calf, nurse shark and clearnose skate. Comp. Biochem. Physiol. 82C:255 257.
- Bodine, A. B., C. A. Luer, S. A. Gangjee and C. J. Walsh. 1989. In vitro metabolism of the pro-carcinogen aflatoxin B1 by liver preparations of the calf, nurse shark, and clearnose skate. Comp. Biochem. Physiol. 94C:447 453.
- Luer, C. A., P. C. Blum and P. W. Gilbert. 1990. Rates of tooth replace¬ment in the nurse shark, Ginglymostoma cirratum. Copeia 1990(1):182 191.
- Carrier, J. C. and C. A. Luer. 1990. A comparison of growth between captive and free nurse sharks, Ginglymostoma cirratum. Copeia 1990(3):686 692.
- Goldstein, L., C. A. Luer and P. C. Blum. 1990. Taurine accumulation by the hearts of embryonic skates, Raja eglanteria. J. Exp. Biol. 150:449 452.
- Crow, G. L., C. A. Luer and C. Barker. 1990. Comparison of serum iron and serum copper levels in captive and wild nurse sharks (Ginglymostoma cirratum). J. Aquaric. Aquat. Sci. 5:79 84.
- Leverone, J. R., C. A. Luer and J. M. Lawrence. 1991. The effect of cations on the specific activities of pyruvate kinase and glucose-6 phosphate dehydrogenase of Luidia clathrata (Say). Comp. Biochem. Physiol. 99B:259 264.
- Bass, N. M., J. A. Manning and C. A. Luer. 1991. Isolation and characterization of fatty acid binding protein in the liver of the nurse shark, Ginglymostoma cirratum. Comp. Biochem. Physiol 98A:355 362.
- Sivak, J. G. and C. A. Luer. 1991. Optical development of the ocular lens of an elasmobranch, Raja eglanteria. Vision Research 31:373 382.
- Luer, C. A. and P. W. Gilbert. 1991. Elasmobranch fish: Oviparous, viviparous and ovoviviparous. Oceanus 34:47 53.
- Kendall, C., S. Valentino, A. B. Bodine and C. A. Luer. 1992. Flow cytometric DNA analysis of nurse shark (Ginglymostoma cirratum) and clearnose skate (Raja eglanteria) peripheral red blood cells. J. Fish. Biol. 41:123-129.
- Goldstein, L., C. A. Luer and P. C. Blum. 1993. Taurine transport characteristics of the embryonic skate (Raja eglanteria) heart. J. Exp. Biol. 182:291-295.
- Conrad, G. W., A. Q. Paulsen and C. A. Luer. 1994. Embryonic development of the cornea in the eye of the clearnose skate, Raja eglanteria. I. Stromal development in the absence of an endothelium. J. Exptl. Zool., 269:263-276.
- Kendall, C., S. Valentino, A. B. Bodine, and C. A. Luer. 1994. Triploidy in a nurse shark (Ginglymostoma cirratum). Copeia 1994(3):825-827.
- Anderson, M. A., C. T. Amemiya, C. A. Luer, R. T. Litman and G. W. Litman. 1994. Complete genomic sequence and patterns of transcription of a member of an unusual family of closely related, chromosomally dispersed immunoglobulin gene clusters in Raja. Internat. Immunol., 6: 1661-1670.
- Takami, S., C. A. Luer and P. P. C. Graziadei. 1994. Microscopic structure of the olfactory organ of the clearnose skate, Raja eglanteria. Anat. Embryol. 190:211-230.
- Pardue, M. T., C. A. Luer, M. G. Callender, B. R. Chou and J. G. Sivak. 1995. The absence of a photopic influence on the refractive development of the embryonic eye of the clearnose skate, Raja eglanteria. Vision Res. 35:1675-78.
- Luer, C. A., C. J. Walsh, A. B. Bodine, J. T. Wyffels and T. R. Scott. 1995. The elasmobranch thymus: Anatomical, histological, and preliminary functional characterization. J. Exptl. Zool., 273:342-354.
- Goldstein, L., E. M. Davis-Amaral, P. C. Blum and C. A. Luer. 1995. The role of anion channels in osmotically activated taurine release from embryonic skate (Raja eglanteria) heart. J. Exp. Biol., 198:2635-2637.
- Rast, J. P., M. K. Anderson, S. Strong, C. Luer, R. T. Litman and G. W. Litman. 1997. α, β, γ and δ T cell antigen receptor genes arose early in vertebrate phylogeny. Immunity, 6:1-11.
- Musch, M. W., C. A. Luer, E. M. Davis-Amaral and L. Goldstein. 1997. Hypotonic stress induces translocation of the osmolyte channel protein pICln in embryonic skate (Raja eglanteria) heart. J. Exptl. Zool., 277:460-463.
- Sisneros, J. A., T. C. Tricas and C. A. Luer. 1998. Response properties and biological function of the skate electrosensory system during ontogeny. J. Comp. Physiol., 183:87-99.
- Walsh, C. J. and C. A. Luer. 1998. Comparative phagocytic and pinocytic activities of leucocytes from peripheral blood and lymphomyeloid tissues of the nurse shark (Ginglymostoma cirratum Bonaterre) and the clearnose skate (Raja eglanteria Bosc). Fish Shellfish Immunol., 8:197-215.
- Hensley, R. A., M. J. McCoid and C. A. Luer. 1998. Litter variation in the Atlantic guitarfish (Rhinobatidae: Rhinobatos lentiginosus) with comments on distribution in the Gulf of Mexico. Southwestern Naturalist. 43:501-504.
- Anderson, M. K., S. J. Strong, R. T. Litman, C. A. Luer, C. T. Amemiya, J. P. Rast and G. W. Litman. 1999. A long form of the skate IgX gene exhibits a striking resemblance to the new shark IgW and IgNARC genes. Immunogenetics, 49:56-67.
- Rasmussen, L. E. L., D. L. Hess and C. A. Luer. 1999. Alterations in serum steroid concentrations in the clearnose skate, Raja eglanteria: Correlations with season and reproductive status. J. Exp. Zool.284:575-585.
- Miracle, A. L., M. K. Anderson, R. T. Litman, C. J. Walsh, C. A. Luer, E. V. Rothenberg and G. W. Litman. 2001. Complex expression patterns of lymphocyte-specific genes during the development of cartilaginous fish implicate unique lymphoid tissues in generating an immune repertoire. Internat. Immunol. 13:567-580.
- Walsh, C. J., J. T. Wyffels, A. B. Bodine and C. A. Luer. 2002. Dexamethasone-induced apoptosis in immune cells from peripheral circulation and lymphomyeloid tissues of juvenile clearnose skates, Raja eglanteria. Dev. Comp. Immunol. 26:623-633.
- Manire, C. A., C. J. Walsh, H. L. Rhinehart, D. E. Colbert, D. R. Noyes and C. A. Luer. 2003. Alterations in blood and urine parameters in Florida manatees, Trichechus manatus latirostris, from simulated conditions of release following rehabilitation. J. Zoo Biol. 22:103-120.
- Anderson, M. K., R. Pant, A. L. Miracle, X. Sun, C. A. Luer, C. J. Walsh, J. C. Telfer, G. W. Litman and E. V. Rothenberg. 2004. Evolutionary origins of lymphocytes: Ensembles of T cell and B cell transcriptional regulators in a cartilaginous fish. J. Immunol. 172:5851-5860.
- Eason, D. D., R. T. Litman, C. A. Luer, W. Kerr and G. Litman. 2004. Expression of individual immunoglobulin genes occurs in an unusual system consisting of multiple independent loci. Eur. J. Immunol. 34:2551-2558.
- Walsh, C. J., C. A. Luer and D. R. Noyes. 2005. Effects of environmental stressors on lymphocyte proliferation in Florida manatees, Trichechus manatus latirostris. Vet. Immunol. Immunopathol. 103:247-256.
- Wyffels, J. T., C. J. Walsh, C. A. Luer and A. B. Bodine. 2005. In vivo exposure of clearnose skates, Raja eglanteria, to ionizing X-radiation: Acute effects on the thymus. Dev. Comp. Immunol. 29(4):315-331.
- Walsh, C. J., J. D. Toranto, C. T. Gilliland, D. R. Noyes, A. B. Bodine and C. A. Luer. 2006. Nitric oxide production by nurse shark (Ginglymostoma cirratum) and clearnose skate (Raja eglanteria) peripheral blood leucocytes. Fish Shellfish Immunol. 20:40-46.
- Walsh, C. J., C. A. Luer, A. B. Bodine, C. A. Smith, H. L. Cox, D. R. Noyes and M. Gasparetto. 2006. Elasmobranch immune cells as a source of novel tumor cell inhibitors: Implications for public health. Integr. Comp. Biol. 46:1072-1081.
- Wyffels, J. T., C. A. Luer, C. J. Walsh and A. B. Bodine. 2007. In vivo exposure of clearnose skates, Raja eglanteria, to ionising X-radiation: Acute effects on the peripheral blood, spleen, and epigonal and Leydig organs. Fish Shellfish Immunol., 23:401-418.
- Luer, C. A., C. J. Walsh, J. T. Wyffels, and A. B. Bodine. 2007. Normal embryonic development in the clearnose skate, Raja eglanteria, with experimental observations on artificial insemination. Env. Biol. Fish., 80 (2-3):239-255.
- Gillis, J. A., M. S. Modrell, R. G. Northcutt, K. C. Catania, C. A. Luer and C. V. H. Baker. 2012. Electrosensory ampullary organs are derived from lateral line placodes in cartilaginous fishes. Development, 139:3142-3146.
- Walsh, C. J., C. A. Luer, J. E. Yordy, T. Cantu, J. Miedema, S. R. Leggett, B. Leigh, P. Adams, M. Ciesla, C. Bennett and A. B. Bodine. 2013. Epigonal conditioned media from bonnethead shark, Sphyrna tiburo, induces apoptosis in a T-cell leukemia cell line, Jurkat E6-1. Marine Drugs, 11:3224-3257.
- Ritchie, K.B., M. Schwarz, J. Mueller, V.A. Lapacek, D. Merselis, C.J. Walsh and C. A. Luer. 2017. Survey of antibiotic-producing bacteria associated with the epidermal mucus layers of rays and skates. Front. Microbiol. 8:1050. doi.org/10.3389/fmicb.2017.01050.
Book Chapters
- Luer, C. A. 1989. Elasmobranchs (sharks, skates, and rays) as animal models for biomedical research. In: “Nonmammalian Models for Biomedical Research.” (A. Woodhead, ed.) CRC Press, Boca Raton, FL, pp. 121 147.
- Luer, C. A., C. J. Walsh and A. B. Bodine. 2004. The immune system of sharks, skates, and rays. In: “Biology of Sharks and Their Relatives.” (J. Carrier, J. Musick and M. Heithaus, eds.) CRC Press, Boca Raton, FL. pp. 369-395.
- Walsh, C. J. and C.A. Luer. 2004. Elasmobranch hematology: Identification of cell types and practical applications. In: “Elasmobranch Husbandry Manual: Proceedings of the First International Elasmobranch Husbandry Symposium, 2001.” (M. Smith, D. Warmolts, D. Thoney and R. Hueter, eds.) Special Publication of the Ohio Biological Survey, Number 16, Columbus, OH. pp. 309-326.
- Luer, C. A., C. J. Walsh and A. B. Bodine. 2012. Recent Advances in Elasmobranch Immunology. In: “Biology of Sharks and Their Relatives.” 2nd Edition. (J. Carrier, J. Musick and M. Heithaus, eds.) CRC Press, Boca Raton, FL. pp., 403-420.
- Luer, C.A., C.J. Walsh and A.B. Bodine. 2015. Sites of Immune Cell Production in Elasmobranch Fishes: Lymphomyeloid Tissues and Organs. In: “Immunobiology of the Shark.” (S Smith, R Sim and M Flajnik, eds.). CRC Press, Boca Raton, FL, 79-88.
- Walsh, C. J. and C.A. Luer. 2015. Leukocyte function in elasmobranch species: Phagocytosis/chemotaxis. In: “Immunobiology of the Shark.” (S Smith, R Sim and M Flajnik, eds.). CRC Press, Boca Raton, FL, 105-122.
- Walsh, C. J. and C.A. Luer. 2015. In Vitro Culture of Elasmobranch Cells. In: “Immunobiology of the Shark.” (S Smith, R Sim and M Flajnik, eds.). CRC Press, Boca Raton, FL, 255-266.
Books Edited
- Development of Non-teleost Fishes. Yvette W. Kunz, Carl A. Luer and B. G. Kapoor, editors. 2009. Science Publishers, Inc. Enfield, NH
-